Purpose:
The method of continuous variations allows scientists to determine the mole ratio of two reactants in a chemical reaction. Typically the mole ratios of reactants and products can be found from a balanced chemical equation, however when the formulas of the products are unknown, experiments can be conducted to discover this ratio. The purpose of this lab is to discover the optimum ratio of the two reactants, meaning that the greatest amount of reactants have been consumed and the greatest amount of product has been formed; this identifies the ratio of moles of reactants in the reaction. Identifying the mole to mole ratio of the reactants allows further stoichiometric calculations to be made.
Data Tables:
Ratio of NaClO to Na2S2O3 (in mL) : Temperature of Mixed Solution (degrees Celsius) after 2 Minutes:
50 : 0 22 degrees C
45 : 5 34 degrees C
40 : 10 41 degrees C
35 : 15 36 degrees C
30 : 20 34.5 degrees C
25 : 25 32 degrees C
20 : 30 29.5 degrees C
15 : 35 28 degrees C
10 : 40 26 degrees C
5 : 45 24.5 degrees C
0 : 50 22 degrees C
50 : 0 22 degrees C
45 : 5 34 degrees C
40 : 10 41 degrees C
35 : 15 36 degrees C
30 : 20 34.5 degrees C
25 : 25 32 degrees C
20 : 30 29.5 degrees C
15 : 35 28 degrees C
10 : 40 26 degrees C
5 : 45 24.5 degrees C
0 : 50 22 degrees C
Graphs:
Conclusion:
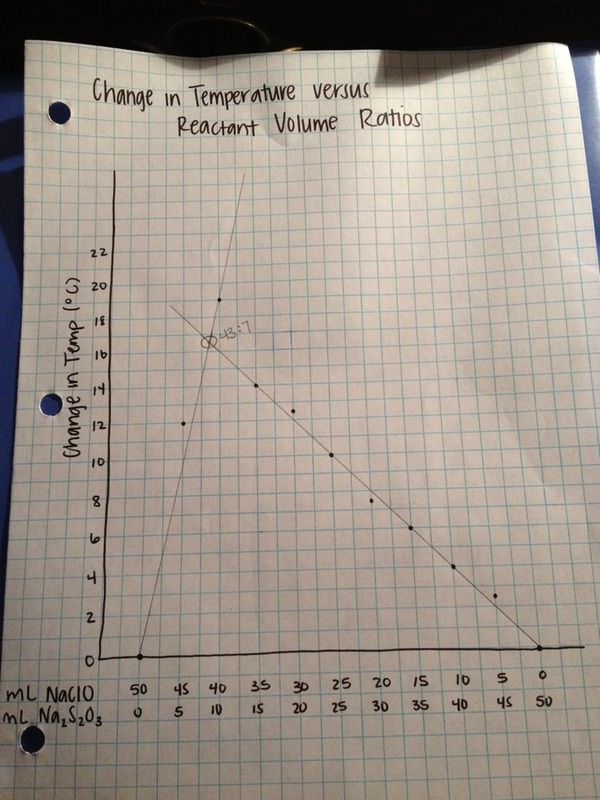
After completing the lab to find the optimum mole ratio of NaClO and Na2S2O3 using the method of continuous variations, it was discovered based on the data that this ratio occured when there was 40 mL of NaClO and 10 mL of Na2S2O3 in the mixture, meaning there is a 4:1 mole ratio, as this produced a temperature change of 19 degrees Celsius from the originial starting temperature of 22 degrees Celsius. When graphing all of the temperature change data and using lines of best fit to find the mole ratio, the point of intersection occured when there was 43 mL of NaClO and 7 mL of Na2S2O3 in the solution. It can be assumed that this is close to a 6:1 mole ratio. This would be the more accurate ratio, as it is reflecting the best fit values of temperature change, and would less likely be affected by any outliers.
Discussion of Theory:
The method of continuous variations allows you to find the mole ratio of reactants. As previously stated, when given a balanced chemical equation the mole ratios of reactants and products is expressed by the coefficients of those formulas. However, the formula of the reactants and products are not always known. When using the method of continuous variations, the total number of moles is kept constant, but the ratios of the reactants are varied. During the chemical reaction, these reactants will produce some sort of measurable property, be it color, a precipitate, or in this instance, heat. This specific experiment involved the occurence of an exothermic reation, so the measurable property was the heat given off and the temperature change of each ratio in comparison with the starting temperature. The heat that is produced is directly proportional to the amount of reaction that is occuring; this means that the temperature change is proportional to the quantity of reactants consumed. When temperature change is found to be at its highest, that identifies that the amount of product formed is at its highest, because this measurable product occurs as a result of maximum consumption of the reactants. In a chemical equation, the stoichiometric ratio will allow the reaction to consume as many reactants as possible and form as much product as capable, so when the measurable property during a continuous variations experiment reflects the same idea, it is clear that this method is effective in finding the mole ratio.
Analysis Questions
1. During the experiment, a constant volume of reactants was kept to ensure that the total number of moles of the reaction was also constant. This ensured that the temperature change had to be a result of the quantity of reactants being consumed, because there would be the same total volume of reactants. There can only be one independent variable in an experiment.
2. The term "limiting reagent" refers to the reactant that is able to be completely consumed in a reaction. This reactant is completely consumed, and it therefore limits the amount of product that can be formed.
3. The precision of the data is limited by the measurement of volume. While the total volume of the solution is kept constant at 50 mL, the volume of each reactant is varying in the different ratios. In each ratio, the amount of reactants available limits the amount of reaction that occurs and the amount of heat that is produced, so if the volume is limited than the data will be less precise, because the ratios are further apart and can not be made at even closer intervals, to produce data that is more precise.
4. Along the upward sloping line of my graph, NaClO is the limiting reactant. Na2S2O3 is the limiting reactant along the downward sloping line of the graph.
5. The method of continous variations could be used by measuring other physical properties, such as the color of a reactant or product, or the mass of a precipiate that forms from a reaction.
6. It is more accurate to use the point of intersection of the two lines of best fit to find the mole ratio rather than the ratio connected to the greatest temperature change because the lines of best fit give you a ratio that is more closely related to all numbers of your data. You can think of that point almost as a median, as you're using the lines to find a ratio that is close to as many data points as possible. The ratio that has the greatest temperature change could be an outlier, taken as a result of an incorrect measurement, or perhaps resulting from a source of error. The best fit gives you a ratio that results from evaluating all the data as a whole, not just choosing the highest value and calling it the best ratio.
2. The term "limiting reagent" refers to the reactant that is able to be completely consumed in a reaction. This reactant is completely consumed, and it therefore limits the amount of product that can be formed.
3. The precision of the data is limited by the measurement of volume. While the total volume of the solution is kept constant at 50 mL, the volume of each reactant is varying in the different ratios. In each ratio, the amount of reactants available limits the amount of reaction that occurs and the amount of heat that is produced, so if the volume is limited than the data will be less precise, because the ratios are further apart and can not be made at even closer intervals, to produce data that is more precise.
4. Along the upward sloping line of my graph, NaClO is the limiting reactant. Na2S2O3 is the limiting reactant along the downward sloping line of the graph.
5. The method of continous variations could be used by measuring other physical properties, such as the color of a reactant or product, or the mass of a precipiate that forms from a reaction.
6. It is more accurate to use the point of intersection of the two lines of best fit to find the mole ratio rather than the ratio connected to the greatest temperature change because the lines of best fit give you a ratio that is more closely related to all numbers of your data. You can think of that point almost as a median, as you're using the lines to find a ratio that is close to as many data points as possible. The ratio that has the greatest temperature change could be an outlier, taken as a result of an incorrect measurement, or perhaps resulting from a source of error. The best fit gives you a ratio that results from evaluating all the data as a whole, not just choosing the highest value and calling it the best ratio.